[Heat and Thermophysics labs]
#1 Gases lab
#2 Specific heat lab
#1 Gases lab
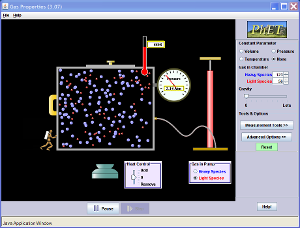
| concept | Boyle's Law shows the relationship between pressure and volume and will have the equation:
P=(N(R)T)/V. Gay-Lussac's law is the relationship between pressure and temperature and will therefore have the equation: P=(N(R))/V * T, and the slope of the graph will be N(kb)/V. Charles' Law shows the relationship between temperature and volume and will therefore have the equation: PV=(N(R))T. |
Equipments | this is ideal gas law experiment-it is almost impossible to work this out in real life. |
| purpose | Students simulate the ideal gas law (PV=NRT).The data that can be collected is based on Boyle's Law, which shows the relationship between volume and pressure, Gay-Lussac's Law, which shows the relationship between temperature and pressure, and Charles' Law, which shows the relationship between temperature and volume. |
| Possible Errors |
|
| simulation |
#2 Specific heat lab
| concept | When heat is transferred between objects, the temperature of the objects change. How much each objects's tempeature changes depends upon how much of the hear energy added or lost goes toward changing the kinetic energy of the object's molcules and how much goes to internal potential energy. Most materials have widely different specific heats. | Equipments | metal spheres ice thermometer tap water styrotoam cup mass scale graduated cylinder |
| purpose | Students measure the specific heat and molar heat capacity of several common metals. |
| Possible Errors |
|
| Simulation |