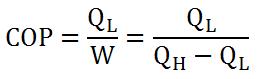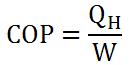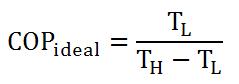Heat Pumps
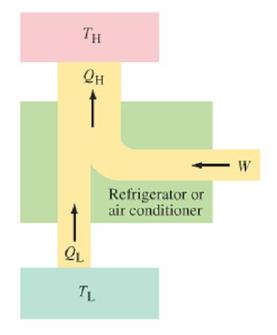
Heat pumps operate like the reverse of heat engines. They transfer heat out of a cool environment into a warm environment (or vice versa) by doing work, as depicted in the diagram to the left. This agrees with Clausius's statement of the 2nd law of thermodynamics, which states that heat only spontaneously flows from a hot object to a cold object.
Like heat engines, heat pumps also have an "efficiency" factor that determines how "good" it is: the coefficient of performance (COP). For refrigerators and air conditioners, COP is determined by equation 1. For heat pumps that deliver warmth or Q(H) in an area, equation 2 is used. COP, unlike efficiency for heat engines, is always greater than one.
Also, like heat engines, there's an ideal heat pump, where Q could be replaced by T. Equation 3 gives the ideal heat pump for refrigerators and air conditioners. For devices that brings warmth from a cold environment to a warmer environment, just switch the T(L) into T(H).
For more information, take a look at the animation at the bottom.
Like heat engines, heat pumps also have an "efficiency" factor that determines how "good" it is: the coefficient of performance (COP). For refrigerators and air conditioners, COP is determined by equation 1. For heat pumps that deliver warmth or Q(H) in an area, equation 2 is used. COP, unlike efficiency for heat engines, is always greater than one.
Also, like heat engines, there's an ideal heat pump, where Q could be replaced by T. Equation 3 gives the ideal heat pump for refrigerators and air conditioners. For devices that brings warmth from a cold environment to a warmer environment, just switch the T(L) into T(H).
For more information, take a look at the animation at the bottom.