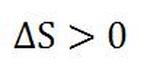Entropy
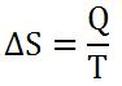
The second law of thermodynamics is also stated in terms of a quantity called entropy. Entropy is a given state of a system, like a given temperature, volume, pressure, and etc. Normally, like potential energy (see Chapter 6), the change in entropy of a system is more important than the absolute amount of entropy at a given time. According to Clausius, the change in entropy is calculated by the equation to the right, where Q is the amount of heat of the system and T is the temperature of the system in degrees Kelvin.
An interesting phenomena regarding entropy is that the total entropy of any system plus that of its environment increases as a result of any natural processes; even though one part of the system may decrease in entropy, another part increases by a greater amount. This means that the total entropy of any isolated system never decreases; it only increases for real processes or stays the same for idealized processes. Thus the inequality below is always true for any real processes. Because of this fact, entropy is one of the factors that never gets conserved during any natural processes, unlike force, work, or energy.