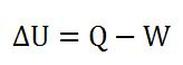The First Law of Thermodynamics
Objectives Covered:
(1) Relate the heat absorbed by a gas, the work performed by the gas, and the internal energy change of the gas for any of the processes above.
(2) Relate the work performed by a gas in a cyclic process to the area enclosed by a curve on a pV diagram.
Definition of the First Law
The first law of thermodynamics is generally stated as the equation below:
Here, Q is net heat added to the system and W is the net work done by the system. In other words, Q is positive is heat is added and negative if heat is lost. Also, W is positive if there's work done by the system or negative if there's work done on the system.
Because the first law of thermodynamics deals with energy conservation, this law is also known as the law of conservation of energy.
Because the first law of thermodynamics deals with energy conservation, this law is also known as the law of conservation of energy.