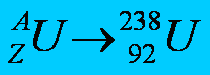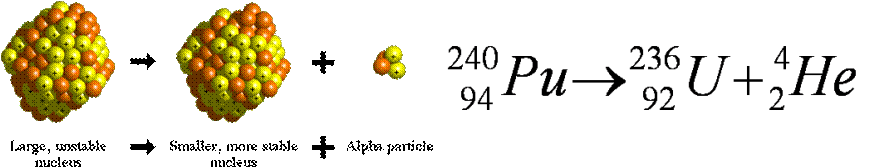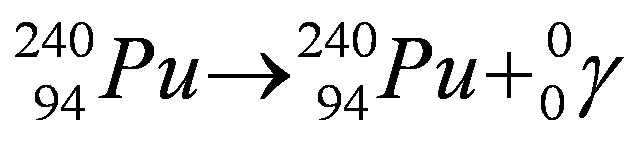Nuclear Physics
Before starting anything, let's review the notation.
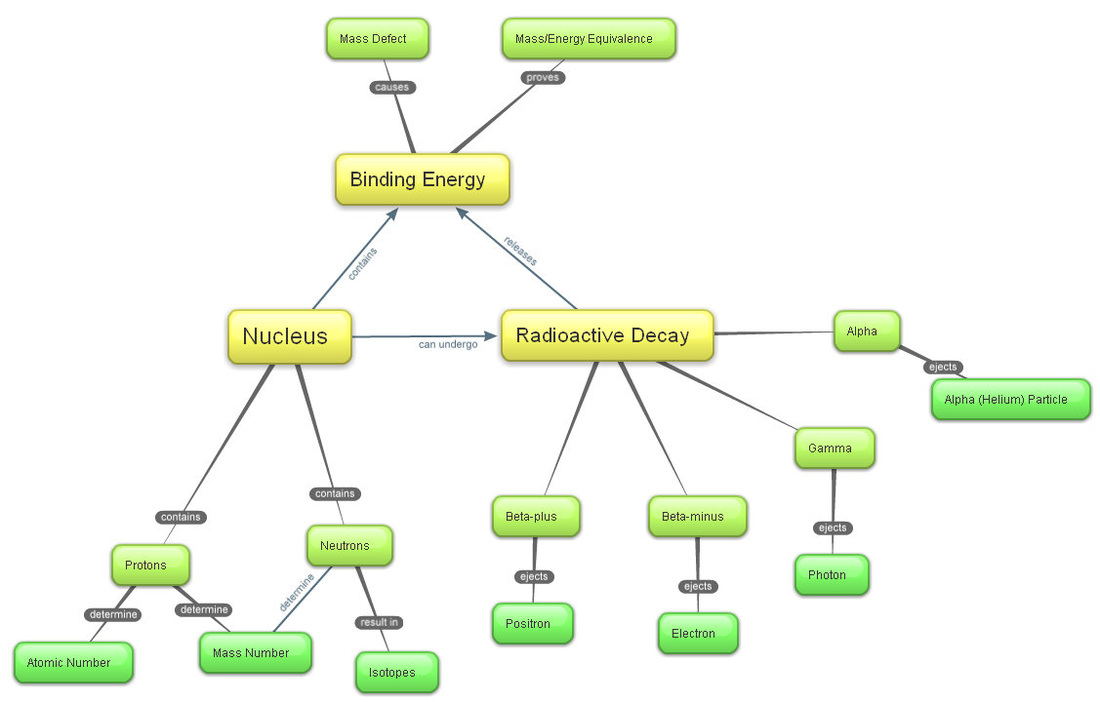
A: The top number, your mass number. It is the total number of protons and neutrons in one atom of an element. Different numbers of neutrons for the same element result in isotopes.
Z: The bottom number, your atomic number. It is the total number of protons in one atom of an element. The number of protons in the atom on one particular element is ALWAYS the same; it is what defines the element.
Z: The bottom number, your atomic number. It is the total number of protons in one atom of an element. The number of protons in the atom on one particular element is ALWAYS the same; it is what defines the element.
Isotopes
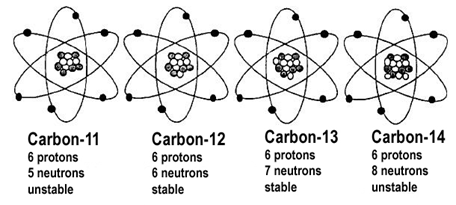
Atoms of the same element that have DIFFERENT mass are called isotopes. This is a result of having extra neutrons.
For example, carbon is always element #6. However, we can differentiate between the isotopes by calling them in terms of their mass.
Eg: Carbon-12, Carbon-14.
Eg: Carbon-12, Carbon-14.
Radioactivity
Radioactive decay occurs when an unstable nucleus releases energy and/or particles. The AP exam only tests 3 (technically 4) types of decay: Alpha, Beta (Plus and Minus), and Gamma decay.
Alpha Decay
In alpha decay, a large, unstable nucleus releases an alpha particle (a helium atom) and becomes a smaller, more stable nucleus.
Beta-Minus Decay
In beta-minus decay (sometimes known simply as beta decay), a nucleus releases an electron while a neutron in the nucleus becomes a proton.
Beta-Plus Decay
In beta-plus decay (sometimes referred to as positron decay), a nucleus releases a positron (which is like an electron, but has a positive charge).
Gamma Decay
In gamma decay, a nucleus releases a photon, a particle of light (also known as a gamma particle).
Tips on Radioactive Decay Processes
Radioactive processes nearly always appear on the multiple choice section of the AP exam. In nearly all cases, the questions simply you to fill in a blank.
For example, they will ask: Radium (atomic number 228, mass number 88) undergoes beta decay. What are the resultant components after the process?
To solve this, you will first need to know what occurs in beta decay (tip: always assume beta decay is beta-minus decay unless told otherwise). Then you will need to be able to do some simply addition and subtraction. The sum of the mass numbers of all components on the left side and the sum of the mass numbers of all components on the right side should be equal, as should be the sum of the atomic numbers. Choose the correct answer, which in this case is an electron and Actinium (atomic number 228, mass number 89).
Basically, all you need to do is be familiar with the processes and make sure the math adds up.
For example, they will ask: Radium (atomic number 228, mass number 88) undergoes beta decay. What are the resultant components after the process?
To solve this, you will first need to know what occurs in beta decay (tip: always assume beta decay is beta-minus decay unless told otherwise). Then you will need to be able to do some simply addition and subtraction. The sum of the mass numbers of all components on the left side and the sum of the mass numbers of all components on the right side should be equal, as should be the sum of the atomic numbers. Choose the correct answer, which in this case is an electron and Actinium (atomic number 228, mass number 89).
Basically, all you need to do is be familiar with the processes and make sure the math adds up.
Mass/Energy Equivalence and Mass Defect
In 1905, Einstein published his theory of Energy-Mass Equivalence, in which he postulated that mass held an enormous amount of energy within itself, called binding energy.
Binding Energy
Binding energy (also referred to as rest mass energy) is an enormous amount of energy present in an atom that holds it together. The binding force is a powerful, short-distance nuclear force that keeps all of the components of an atom together. It is not gravitation, electric, or chemical.
Mass Defect
The mass of an atom and the sum of the masses of its components is different, with the masses of the components being greater.
For example, the mass of carbon-12 is 11.996709 amu (1 amu = 1.660559 x 10^-27 kg, approximately the mass of a proton or neutron), where as the mass of its components, 6 protons and 6 neutrons, is 12.095646 amu. There is a mass defect of 0.098937 amu.
The extra, "lost mass" of the individual components is converted into binding energy to hold the atom together.
Mass-Energy Equivalence
Based on these experimental findings, Einstein postulated that mass and energy could be turned into each other. He came up with perhaps the most famous formula in all of science, to explain how it works. You can check the extras section for a recording of him explaining his findings.