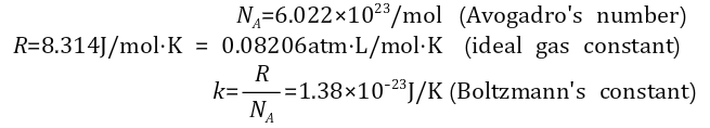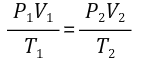Temperature and Kinetic Theory
AP Objectives
These are all the objectives stated in the AP course description that relate to thermal expansion, ideal gases, and kinetic theory:
A. Thermal expansion (~2% of exam)
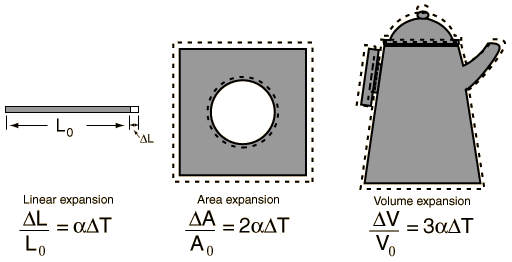
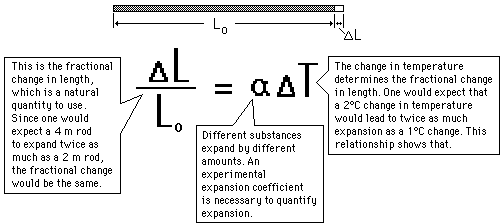
a) Analyze what happens to the size and shape of an object when it is heated.
B. Kinetic theory and thermodynamics (~7% of exam)
1. Kinetic model
a) State the assumptions of the model.
b) State the connection between temperature and mean translational kinetic
energy, and apply it to determine the mean speed of gas molecules as a function
of their mass and the temperature of the gas.
c) State the relationship among Avogadro’s number, Boltzmann’s constant, and
the gas constant R, and express the energy of a mole of a monatomic ideal gas
as a function of its temperature.
d) Explain qualitatively how the model explains the pressure of a gas in terms of
collisions with the container walls, and explain how the model predicts that, for
fixed volume, pressure must be proportional to temperature.
2. Ideal gases
a) Relate the pressure and volume of a gas during an isothermal expansion or
compression.
b) Relate the pressure and temperature of a gas during constant-volume heating or
cooling, or the volume and temperature during constant-pressure heating or
cooling.
c) Calculate the work performed on or by a gas during an expansion or
compression at constant pressure.
d) Understand the process of adiabatic expansion or compression of a gas.
e) Identify or sketch on a PV diagram the curves that represent each of the above
processes.
A. Thermal expansion (~2% of exam)
a) Analyze what happens to the size and shape of an object when it is heated.
B. Kinetic theory and thermodynamics (~7% of exam)
1. Kinetic model
a) State the assumptions of the model.
b) State the connection between temperature and mean translational kinetic
energy, and apply it to determine the mean speed of gas molecules as a function
of their mass and the temperature of the gas.
c) State the relationship among Avogadro’s number, Boltzmann’s constant, and
the gas constant R, and express the energy of a mole of a monatomic ideal gas
as a function of its temperature.
d) Explain qualitatively how the model explains the pressure of a gas in terms of
collisions with the container walls, and explain how the model predicts that, for
fixed volume, pressure must be proportional to temperature.
2. Ideal gases
a) Relate the pressure and volume of a gas during an isothermal expansion or
compression.
b) Relate the pressure and temperature of a gas during constant-volume heating or
cooling, or the volume and temperature during constant-pressure heating or
cooling.
c) Calculate the work performed on or by a gas during an expansion or
compression at constant pressure.
d) Understand the process of adiabatic expansion or compression of a gas.
e) Identify or sketch on a PV diagram the curves that represent each of the above
processes.
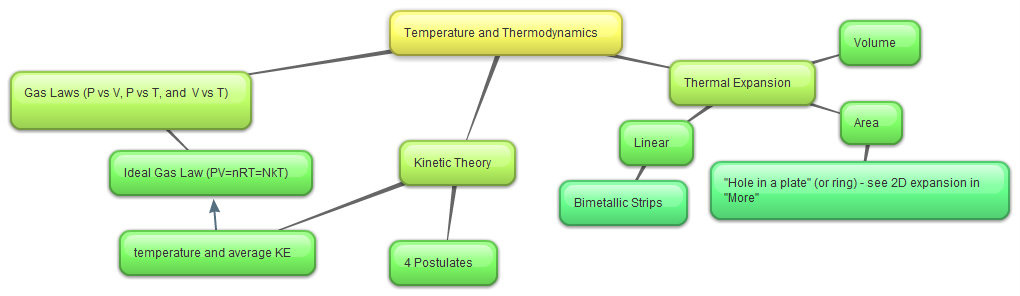
Summary
Constants
Moles
1 mol = 6.022*10^23 (atoms/molecules)
1 mol of Carbon-12 is defined to have a mass of 12g
Therefore, the mass of 1 mol of a certain atom/molecule has a mass equal to its atomic mass in grams.
1 mol of Carbon-12 is defined to have a mass of 12g
Therefore, the mass of 1 mol of a certain atom/molecule has a mass equal to its atomic mass in grams.
Formulas
1. Thermal Expansion
2. Gas Laws
STP - Standard Temperature and Pressure is defined to be at 1 atm (101325 Pa) and 273.15K
The ideal gas law can also be written as (when only changing P, V, and T for the same gas):
The ideal gas law can also be written as (when only changing P, V, and T for the same gas):
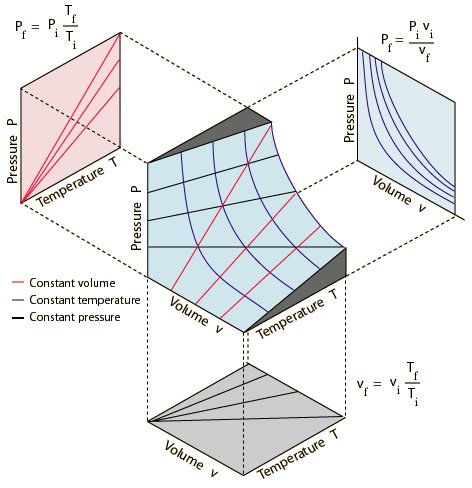
Visualization of P vs V, P vs T, and V vs T graphs (and all 3 at the same time)
3. Kinetic Theory
Postulates of Kinetic Theory
KT assumes the following:
KT assumes the following:
- There are many molecules moving in random directions at various speeds.
- The molecules are far apart from each other.
- The molecules only interact when they collide (no EM or gravitational forces).
- Collisions (with other molecules and the walls) are perfectly elastic.
KT Derivation
Practice
| Practice Questions | |
| File Size: | 55 kb |
| File Type: | doc |
| Answer Sheet | |
| File Size: | 83 kb |
| File Type: | doc |
AP Questions for the detail oriented
More
Thermal Expansion (includes 2D expansion)
Thermal Expansion Lesson
Gas Laws Refresher
Gas Molecules Simulation
Gas Law - P vs V
Boyle's Law Demo
Steven Wang